There’s more about entropy at NASA and Physics World too. Want to know why time might flow in one direction? Have you ever thought about the time before the Big Bang? The entire entropy concept plays an important role in understanding them. You can read more about entropy here in Universe Today. To give you some relief, not everyone involved in the study of cosmology is totally in agreement with entropy’s so-called role in the grand scheme of things though. 
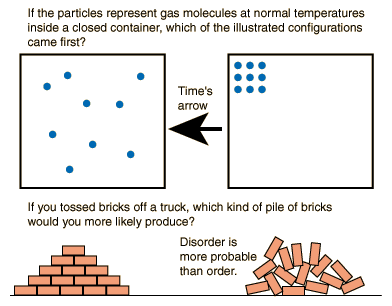
a heat death, wherein energy can no longer be extracted from anymore. So what? Well, also just like all isolated systems, the universe is therefore also expected to end up in a useless heap in equilibrium, a.k.a. The entropy change for this reaction is highly negative because three gaseous molecules are being converted into two liquid. Thus, like in all isolated systems, the entropy of the universe is expected to be increasing. The entropy change for the formation of liquid water from gaseous hydrogen and oxygen can be calculated using this equation: 2H 2(g) + O 2(g) 2H 2O(l) So 2(69.9) 2(131.0) + 1(205.0) 327 J/K mol. As such, it should also be governed by the second law of thermodynamics. Since the concept of entropy applies to all isolated systems, it has been studied not only in physics but also in information theory, mathematics, as well as other branches of science and applied science.īecause the accepted view of the universe is that of one that is finite, then it can very well be considered as a closed system. Without it, like when maximum entropy has already been achieved, there is no way that work can be performed. Notice, however, that work can only be done for as long as there is a difference in temperature. The measurement of the extent of this evening-out process is called entropy.ĭuring the process of attaining equilibrium, it is possible to tap into the system to perform work, as in a heat engine. In both cases, the physical quantities which started to be uneven between the two bodies/regions even out in the end, i.e., when equilibrium is achieved. This goes on until the pressures in the adjacent regions even out. Thus, when the fluid, air in this case, comes rushing in, they do so in the form of strong winds. This happens because all fluids flow from a region of high pressure to a region of low pressure. Ever heard of a low pressure area? It’s what weather reporters call a particular region that’s characterized by strong winds and perhaps some rain. However, after sufficient time has passed, the system reaches a uniform color, a state much easier to describe and explain.īoltzmann formulated a simple relationship between entropy and the number of possible microstates of a system, which is denoted by the symbol Ω.Second example. The dye diffuses in a complicated manner, which is difficult to precisely predict. 
However, this description is relatively simple only when the system is in a state of equilibrium.Įquilibrium may be illustrated with a simple example of a drop of food coloring falling into a glass of water. Therefore, the system can be described as a whole by only a few macroscopic parameters, called the thermodynamic variables: the total energy E, volume V, pressure P, temperature T, and so forth. The ensemble of microstates comprises a statistical distribution of probability for each microstate, and the group of most probable configurations accounts for the macroscopic state. C in Eq.2.3 is an undetermined constant which is unimportant since we are always interested in changes in entropy, so it is convenient to set C 0. The large number of particles of the gas provides an infinite number of possible microstates for the sample, but collectively they exhibit a well-defined average of configuration, which is exhibited as the macrostate of the system, to which each individual microstate contribution is negligibly small. The use here of the notation S instead of S indicates that Boltzmann’s expression is valid for any size change of entropy. The collisions with the walls produce the macroscopic pressure of the gas, which illustrates the connection between microscopic and macroscopic phenomena.Ī microstate of the system is a description of the positions and momenta of all its particles. At a microscopic level, the gas consists of a vast number of freely moving atoms or molecules, which randomly collide with one another and with the walls of the container. The easily measurable parameters volume, pressure, and temperature of the gas describe its macroscopic condition ( state). A useful illustration is the example of a sample of gas contained in a container. Ludwig Boltzmann defined entropy as a measure of the number of possible microscopic states ( microstates) of a system in thermodynamic equilibrium, consistent with its macroscopic thermodynamic properties, which constitute the macrostate of the system. Main article: Boltzmann's entropy formula 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
